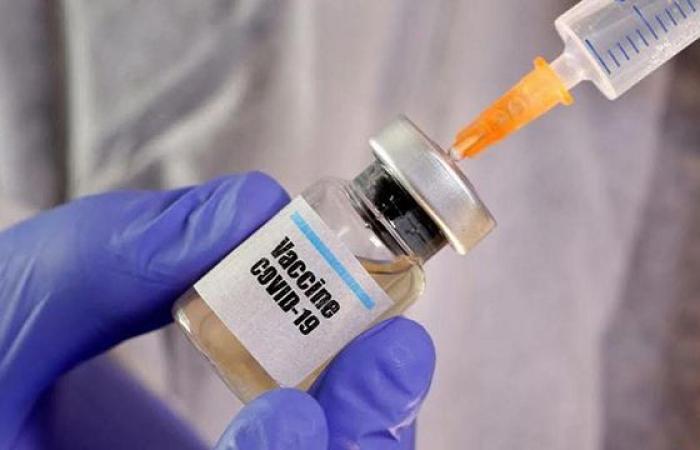
US companies Pfizer and Moderna plan to seek authorization for their Covid-19 vaccines by the end of November in the United States, which would mark an absolute speed record for vaccine development, nine months after the first death from coronavirus in the country.
But the eventual launch of vaccination by the New Year will be too limited to contain the epidemic on its own, which is in its third rebound in the United States, with an alarming rise in contaminations, hospitalizations and deaths – rebound all the more rapid as the wave left a high plateau, the country of Donald Trump having never succeeded in stemming the circulation of the virus.
The US president had been hoping for a vaccine before the November 3 election, but the industry agreed to wait a few more weeks, at the request of health officials, to detect possible serious side effects among the tens of thousands of participants in clinical trials. Trials should answer two questions: Does the vaccine protect? And is it safe?
Albert Bourla, CEO of Pfizer, a partner of the German company BioNTech, announced on Friday that he expected evidence of efficacy by the end of October, but that he would wait until the third week of November to file with the Agence des drugs (FDA) an emergency authorization request, “assuming the data are positive”.
The schedule matches that planned by Moderna, a biotechnology company, which is counting on November 25.
Pfizer and Moderna received $ 1.95 billion and $ 2.48 billion respectively in public money, and they bet on the same new technology, messenger RNA. The method involves inserting genetic instructions into the body to trigger the production of a protein identical to that of the coronavirus (its recognizable spike), and elicit an immune response.
This response was seen in initial trials with volunteers, but the real test is to see if vaccinated people are actually avoiding Covid-19, in real life.
They are each made in two doses spaced three or four weeks apart, and have one drawback: the doses must be stored at very low temperature.
Each company launched its trials at the end of July, with 30,000 participants, half of whom received the vaccine and the other a placebo, blind: they will be considered effective if the number of vaccinated participants falling ill with Covid-19 is reduced by at least 50% compared to the placebo group.
Ten experimental vaccines are in the final stages of testing worldwide, but these two are the only ones currently active in the United States.
The first peak in the United States was in April, when the epidemic hit New York and the Northeast hard. With the closure of the economy, the curve fell again but too rapid deconfinement caused a second wave in July, centered on the South and the West.
During the summer and until September, the epidemic declined, but the trend is now clear: Thursday, the number of new cases exceeded 60,000, and several states recorded records.
37,000 people are hospitalized and 950 deaths were recorded Thursday, according to the Covid Tracking Project. Compared to France, and compared to the population, the United States has fewer new cases but more deaths per day.
The North and the Midwest are today the most affected, but the whole country is on alert, including New York, yet a model city for the caution of its deconfinement.
“The contaminations seem to occur during gatherings inside people’s homes, not just in bars and restaurants like this summer,” Thomas Tsai, researcher at Harvard, told AFP.
This was the fear long expressed by epidemiologists: as temperatures cool, people spend more time indoors, where the virus is more easily transmitted.
As for the possible arrival of a vaccine, it will take many months to vaccinate the 330 million Americans. Masks, distancing and possible reconfigurations will remain the most effective weapons to get through the winter.
With MAP
*We just want readers to access information more quickly and easily with other multilingual content, instead of information only available in a certain language.
*We always respect the copyright of the content of the author and always include the original link of the source article.If the author disagrees, just leave the report below the article, the article will be edited or deleted at the request of the author. Thanks very much! Best regards!
These were the details of the news Two vaccines could be ready to be authorized in November in... for this day. We hope that we have succeeded by giving you the full details and information. To follow all our news, you can subscribe to the alerts system or to one of our different systems to provide you with all that is new.
It is also worth noting that the original news has been published and is available at en24news and the editorial team at AlKhaleej Today has confirmed it and it has been modified, and it may have been completely transferred or quoted from it and you can read and follow this news from its main source.