In this groundbreaking study, the Bristol Faculty of Life Sciences research groups selected Professor Peter Cullen of the School of Biochemistry. Dr. Yohei Yamauchi, Associate Professor and Virologist at the School of Cellular and Molecular Medicine.
Dr. Boris Simonetti, a lead researcher at Cullen’s lab, used multiple approaches to find that SARS-CoV-2 recognizes a protein called neuropilin-1 on the surface of human cells to facilitate virus infection.
Yohei, Boris and Pete stated, “When we looked at the sequence of the SARS-CoV-2 spike protein, we found that there was a small sequence of amino acids that appeared to mimic a protein sequence found in human proteins, that were made with interaction Neuropilin-1.
Advertisements
This led us to a simple hypothesis: Could the spike protein from SARS-CoV-2 associate with Neuropilin-1 to aid in viral infection of human cells? Excitingly, we were able to use a number of structural and biochemical approaches to determine that the spike protein from SARS-CoV-2 actually binds to Neuropilin-1.
“After we discovered that the spike protein was bound to Neuropilin-1, we were able to show that the interaction serves to increase SARS-CoV-2 invasion in human cells grown in cell culture.
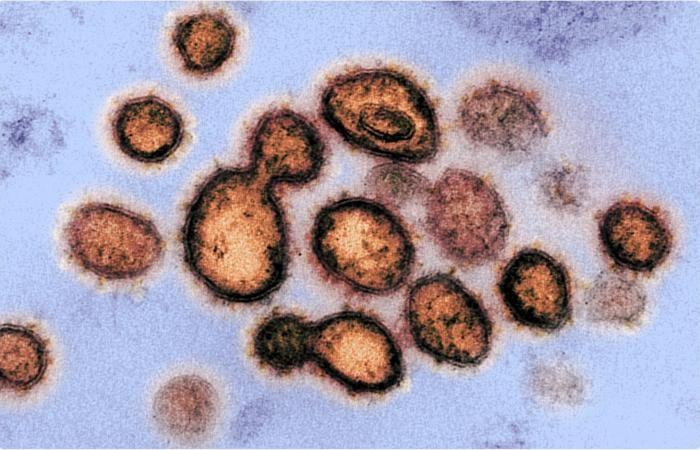
This transmission electron microscope image shows SARS-CoV-2, the virus that causes COVID-19 and was isolated from a patient in the United States. Virus particles appear on the surface of cells that have been cultured in the laboratory. The spines on the outer edge of the virus particles give the coronavirus its name, crown-like. Image was taken and colored in at NIAID’s Rocky Mountain Laboratories (RML) in Hamilton, Montana. Image Credit: NIAID
Die Spike Cleavage Site
The COVID-19 pandemic has caused an immense number of cases exceeding 42.9 million and causing well over a million deaths worldwide. The viral agent SARS-CoV-2 enters the host cell via its spike protein, which binds to the ACE2 receptor of the cell. The spike protein comprises two subunits, S1 and S2, and has a polybasic furin cleavage site at the interface of the two. This allows a host cell proprotein convertase, or furin, to cleave the tip once it has activated the receptor.
The S1 and S2 subunits are still not covalently associated, and the S2 appears to be further primed by the serine protease TMPRSS2. The furin cleavage of the spike protein leads to increased viral infectivity. Furin inhibition or deletion of the polybasic RRAR site reduces virus entry.
The neuropilin factor
The S1 subunit has a C-terminal amino acid sequence that follows the C-end rule (CendR). Peptides of this type bind to Neuropilin-1 (NRP1) and NRP2, which are transmembrane receptors that affect many different processes in the body. The current study aimed to examine the relationship between the two.
The researchers used a GFP-labeled S1 construct that was expressed in culture in ACE2 positive cells. They found that the S1 protein was bound to both ACE2 and NRP-1 in the GFP-S1 cells, but not to the GFP-S1-ΔRRAR cells, which had a deletion of the furin cleavage site. Similar binding of S1 was also observed for NRP2. Some binding to the deletion mutant was also observed, indicating that the neuropilins interacted with the S1 independently of the CendR residues.
The absence of NRP1 reduces infectivity
In cells lacking NRP1 but expressing ACE2, exposure to whole virus was found to result in lower levels of infection. Cells that were missing both were not infected at all. In another cell assay, treatment with the suppressor shRNA, which prevented NRP1 expression, again reduced the level of infection of the virus. The absence of this protein still allows the virus to attach to the cell surface, but its internalization is cut in half. It is thus shown that NRP1 is essential for the virus to enter and infect the cell. Their results also supported the existence of a CendR-independent mechanism for NRP1-mediated elevated infection.
SARS-CoV-2 infection was significantly greater in NRP-positive cells, but no increase was observed in the presence of the T316R mutation, suggesting that this change decreased the association of NRP1 with the S1 subunit. They found that monoclonal antibodies that target the NRP1 protein bound to the CendR binding pocket are expressed on the cell with high specificity and reduce the ability of SARS-CoV-2 to infect the cell. Incubation with soluble ACE2 also prevented SARS-CoV-2 infection, which indicates the validity of the previous finding.
The use of a low molecular weight NRP1 inhibitor EG00229 also showed that it can effectively inhibit S1-NRP1 binding by binding to the b1-CendR binding pocket of NRP1. This therefore represents a therapeutic goal in order to reduce the infectivity of the virus in human cells.
How Neuropilins Increase Viral Infectivity
The viral entry is preceded by spike priming by host proteases. The CendR sequence generated by cleaving the spike protein at the S1 / S2 interface binds to the neuropilins on the cell surface. This is not essential for virus adhesion to the cell surface, but it does improve virus entry and cell infection.
Researchers aren’t sure how neuropilins cause increased levels of viral infection. However, they believe this may be due to the known ability of neuropilins to mediate macropinocytosis, in which the cell engulfs a large pocket of cell membrane and the attached molecules and particles that allow CendR-linked molecules to enter the cell to penetrate.
Both NRP1 and NRP2 are known to be upregulated in lung tissue samples from COVID-19 patients. Therefore, the binding of NRP1 to the S1-CendR motif may be a reason why SARS-CoV-2 is more infectious than SARS-CoV. This can be targeted in future treatments for this disease.
These were the details of the news. A new route for the SARS-CoV-2 entry for this day. We hope that we managed to give you all the details and information. To keep up with all of our news, you can subscribe to the notification system or one of our various systems to receive everything new.
It’s also worth noting that the original news has been published and is available at de24.news. AlKhaleej Today’s editorial team has confirmed this and it has been changed and it may have been fully retransmitted or quoted and you can read and follow this news from its main source.
These were the details of the news A new route for the SARS-CoV-2 entry for this day. We hope that we have succeeded by giving you the full details and information. To follow all our news, you can subscribe to the alerts system or to one of our different systems to provide you with all that is new.
It is also worth noting that the original news has been published and is available at de24.news and the editorial team at AlKhaleej Today has confirmed it and it has been modified, and it may have been completely transferred or quoted from it and you can read and follow this news from its main source.