The most important things at a glance
- In Bern today, federal experts will once again be providing information on the current situation in the pandemic.
- The Swiss regulatory authority Swissmedic is examining the possible Covid-19 vaccine from Pfizer and Biontech. Admission would be conceivable within 100 days.
- The US health authority allows the method of treating Covid-19 patients with antibodies as an emergency application.
- US President Donald Trump criticizes the announcement of promising results in the search for a vaccine a few days after the election as politically motivated.
7.26 a.m.: Lonza is said to produce vaccine candidate
When looking for a vaccine against the coronavirus, Lonza also reports news: The Basel pharmaceutical company will produce a possible vaccine for the US group Altimmune. This is the drug Adcovid, which is administered once via a nasal spray.
Altimmune hopes to bring the vaccine to market in 2021, both companies announced on Tuesday. A clinical study is to be launched in the final quarter of 2020, the results of which should be available in the first quarter of 2021.
Altimmune, based in the state of Maryland, expects the vaccine could protect people from Covid-19 for up to a year or more. In addition, the vaccine should be able to be transported without securing a cold chain.
Lonza is already a manufacturing partner for Moderna’s vaccine. The federal government had already signed a contract with Moderna in August for the purchase of 4.5 million vaccine doses.
7.10 a.m.: Cantons want to go back to the bus regime
In spring, the police could speak a fine for violations of the Corona measures. Since the “extraordinary situation” no longer applies, this is no longer possible – sanctions can only be imposed by filing a criminal complaint.
The cantons would like to be able to distribute fines again: “For enforcement, it is important that minor violations can be punished in the administrative fines procedure,” wrote the cantonal health directors’ conference (GDK) in a statement to the Federal Council that was available to “Blick”.
GDK General Secretary Michael Jordi told the newspaper that one would not want to distribute busses in rows: “Not everyone who does not wear a mask should receive a fine right away,” says Jordi. “With the bus option, however, the police could exert measured pressure and convince those affected that the rules were being followed.”
The Federal Office of Public Health points out that a return to the fines regime would first require an amendment to the Ordinance Fines Act: “This is not planned at the moment.”
Image: Keystone/Anthony Anex
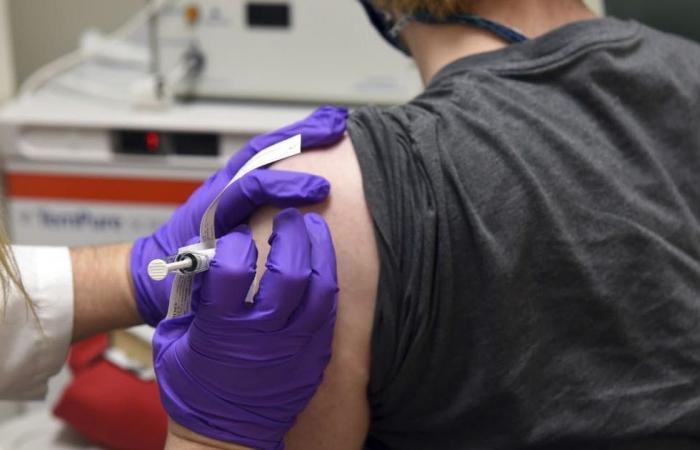
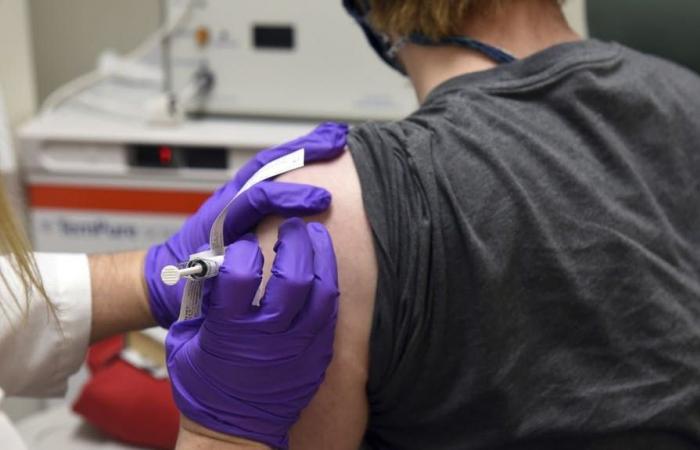
6.14 a.m.: Swissmedic is testing Pfizer Biontech vaccine
The Swiss approval authority Swissmedic is already examining the Covid-19 vaccine candidate with the awkward name “BNT162 b2”, which made headlines on Monday and caused price jumps on the stock exchanges. The developers, the US pharmaceutical company Pfizer and the German immunotherapy specialist Biontech, have submitted an application in Switzerland. “I even think that we can issue an authorization faster than in 100 days,” says Swissmedic Vice Director Philippe Girard in an interview with the “Tages-Anzeiger”. The prerequisite is that all data are available. According to Girard, applicants only submit these in a fast-track process gradually.
The Swissmedic Vice Director said that approval this year would be unthinkable, but it would be possible at the beginning of 2021. Switzerland has not placed a pre-order for the Pfizer Biontech product under development, but has reserved possible vaccines from two other companies. According to the “Tages-Anzeiger”, the distribution struggle is likely to intensify.
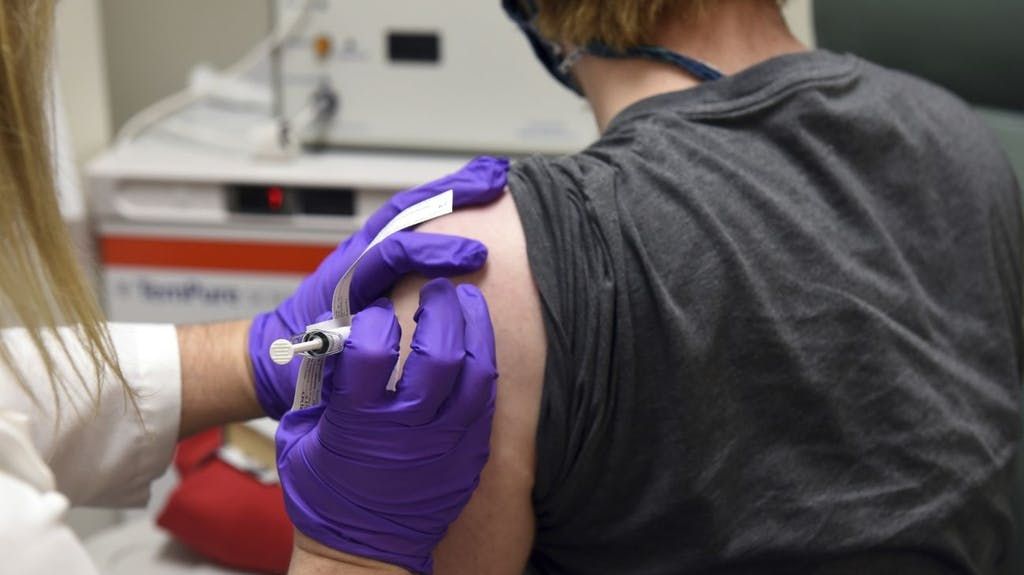
Image: Keystone/University of Maryland School of Medicine via AP
Biontech and Pfizer were the first western manufacturers to publish promising – but limited in the scope of the details – results on Monday. Accordingly, your vaccine offers more than 90 percent protection against Covid-19. The companies want to apply for emergency approval from the FDA starting next week.
5.30 a.m.: FDA grants emergency approval for treatment
The US health authority FDA allows the method developed by the US pharmaceutical company Eli Lilly to treat Covid-19 patients with antibodies as an emergency application. The FDA announced on Monday evening.
The approval is based on clinical studies that have shown that treatment with the agent “Bamlanivimab” reduces the need for hospital stays for Covid-19 patients. It can be used to treat mild to moderate illnesses in adults and children over the age of twelve.

Image: Keystone/David Morrison/Eli Lilly via AP, File
5 a.m .: Trump complains about timing of vaccine news
The incumbent President Donald Trump described the announcement of promising results from the manufacturers of a corona vaccine a few days after the US election as politically motivated. The pharmaceutical company Pfizer “did not have the courage” to announce the good news before the election, criticized Trump on Monday evening (local time) on Twitter. The US company is working on the vaccine with the German company Biontech.
Trump also criticized the Food and Drug Administration (FDA) of his administration, which is responsible for approval in his tweet. The FDA should have announced the results earlier, he complained. “Not for political reasons, but to save lives,” wrote Trump. During the election campaign, he had repeatedly promised that there would be good news on vaccines before the vote – and apparently hoped for a political tailwind from it.
“The FDA and the Democrats didn’t want me to have a vaccine success before the election, so it came out five days later,” Trump wrote on Twitter. His administration’s FDA is headed by Stephen Hahn, whom Republican Trump himself appointed.
BioNTech plans approval application for corona vaccine
Numerous governments have pre-secured millions of doses of the vaccine.
09.11.2020
4 a.m.: Shortly before application for vaccine: Biontech presents figures
Shortly after the publication of encouraging interim results on its corona vaccine, the Mainz-based pharmaceutical company Biontech today presents third-quarter figures. In addition, Biontech wants to give “information on operational progress” in a conference call at 2 pm.
Biontech and the US pharmaceutical company Pfizer were the first western manufacturers to publish promising results of a vaccine study that is crucial for approval on Monday. Your vaccine offers more than 90 percent protection against Covid-19, the two companies said. Serious side effects were not found.
3.31 a.m.: Federal experts provide information today
In Bern today, federal experts will once again be providing information on the current situation in the pandemic. Last weekend, the number of new infections fell slightly. Over 17,000 people were infected with the coronavirus within three days. A total of 169 people died from the lung disease Covid-19. Health Minister Alain Berset warned against too much euphoria. It is premature to see any trend. The situation remains serious. The situation in the intensive care units of the hospitals continues to cause the greatest concern.
2.46 a.m.: How does Hotelplan get through the pandemic?
The tour operator Hotelplan presented its outlook for winter business and the forecasts for summer 2021 today, Tuesday. The pandemic is likely to have continued to have deep marks in international business. Many countries, including Germany and France, still have strict travel restrictions. In contrast, the Swiss are likely to book lively holidays in Switzerland, especially with a view to the coming ski season. The crisis in tourism due to the pandemic is likely to drag on and also affect summer business in 2021. Above all, cross-border tourism is down to earth. A recovery is not expected until 2022 at the earliest.
2 a.m.: Biden win and vaccine push Dow to a record high
The victory of Democrat Joe Biden in the US presidential election and a possible breakthrough in the search for a vaccine against the coronavirus set new records on the US stock exchanges at the beginning of the week. The leading index Dow Jones Industrial and the market-wide S&P 500 soared to highs in the first few minutes of trading on Monday. Stocks that had suffered particularly from the pandemic saw price fireworks.
At the closing bell, the Dow Jones Industrial posted a gain of 2.95 percent to 29,157.97 points. The leading index had just barely missed the jump over the 30,000 mark. Before regular trading, the Dow was temporarily indexed above this level. The S&P 500 rose 1.17 percent to 3550.50 points. The technology-heavy Nasdaq 100, on the other hand, lagged noticeably with a minus of 2.16 percent to 11,830.39 points.
Back to the home page
*We just want readers to access information more quickly and easily with other multilingual content, instead of information only available in a certain language.
*We always respect the copyright of the content of the author and always include the original link of the source article.If the author disagrees, just leave the report below the article, the article will be edited or deleted at the request of the author. Thanks very much! Best regards!
These were the details of the news Switzerland is testing Pfizer Biontech vaccine in an urgent procedure –... for this day. We hope that we have succeeded by giving you the full details and information. To follow all our news, you can subscribe to the alerts system or to one of our different systems to provide you with all that is new.
It is also worth noting that the original news has been published and is available at en24news and the editorial team at AlKhaleej Today has confirmed it and it has been modified, and it may have been completely transferred or quoted from it and you can read and follow this news from its main source.