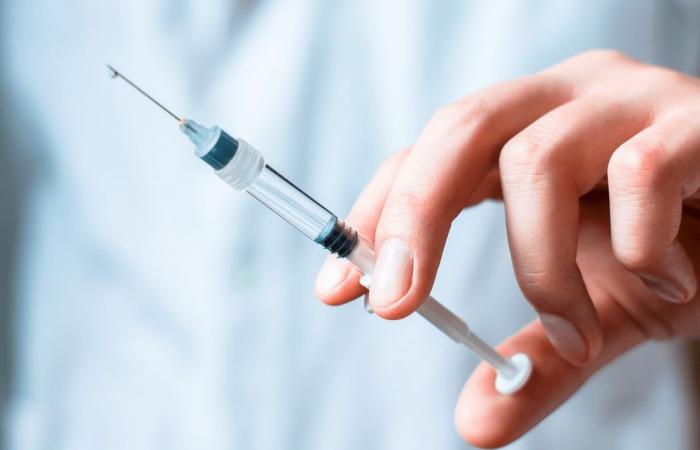
On Friday, the European Medicines Agency gave the green light for an injectable treatment for AIDS, which sets a precedent that can change the lives of millions of people with HIV or seropositive, according to what was published by “The French”.
The treatment consists of a combination of two antiretrovirals, and it can be given to the patient once every month or two, instead of taking daily medication pills to contain the disease, according to what the agency explained in a statement, noting that these two antiretrovirals are the “first” that have been found to have an effect. ” Long term “by injection.
She explained that “the patients will receive intramuscular injections once every month or two instead of the daily medicine tablets.”
The agency’s recommendation to allow this treatment to be marketed must obtain approval from the European Commission in order for it to be put on the market in the EU’s 27 countries.
The agency indicated that the combination of the two antiretroviral drugs Rilpivirin and Capotegravir, which will be sold under the names “Recampase” and “Vocabria”, helps in “preventing the ability of the virus to reproduce.”
The number of people living with AIDS in the world reached 38 million in 2019, including 2.3 million in Europe, according to the World Health Organization.
There is no cure for this disease yet, but antiretrovirals can control virus progression and help prevent transmission.
These were the details of the news A green light in Europe for an injectable treatment for AIDS for this day. We hope that we have succeeded by giving you the full details and information. To follow all our news, you can subscribe to the alerts system or to one of our different systems to provide you with all that is new.
It is also worth noting that the original news has been published and is available at saudi24news and the editorial team at AlKhaleej Today has confirmed it and it has been modified, and it may have been completely transferred or quoted from it and you can read and follow this news from its main source.