Named BBIBP-CorV, another Chinese vaccine proved to be safe and triggered immune responses after initial and intermediate stage tests in humans edit

Sputnik – The race to get a vaccine against Covid-19 won a new strong candidate, according to a statement from Lancet magazine released on Thursday (15).
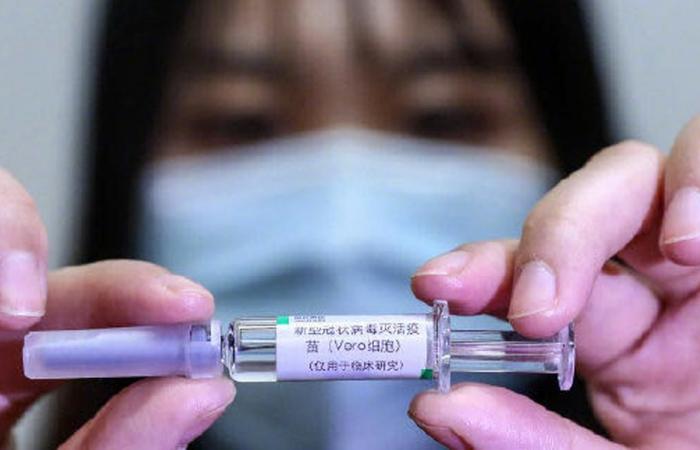
BBIBP-CorV is being developed by the Beijing Institute of Biological Products, a subsidiary of the National Biotechnology Group of China (CNBG).
It has even been approved for an emergency vaccination program in China, aimed at essential workers and other groups of people who face high risk of infection.
BBIBP-CorV includes a mixture of the killed virus with another component, aluminum hydroxide, called an adjuvant because it is known to increase immune responses.
To prove the vaccine’s effectiveness, the final phase three tests, which are ongoing outside China, are still awaited.
The Lancet publication points out that this Chinese vaccine, like the other, did not cause serious side effects, but only mild adverse reactions, such as fever and pain at the injection site.
The results released on Thursday (15) came from a combined trial between phases one and two, involving more than 600 healthy adults and conducted between April 29 and July 30.
You who made it this far, thank you very much for valuing our content. Unlike corporate media, Brasil 247 and TV 247 are financed through their own community of readers and viewers. You can support TV 247 and the Brasil 247 website in several ways. See how in brasil247.com/apoio
These were the details of the news Human tests show another Chinese vaccine promises to be effective against... for this day. We hope that we have succeeded by giving you the full details and information. To follow all our news, you can subscribe to the alerts system or to one of our different systems to provide you with all that is new.
It is also worth noting that the original news has been published and is available at time24.news and the editorial team at AlKhaleej Today has confirmed it and it has been modified, and it may have been completely transferred or quoted from it and you can read and follow this news from its main source.