The ongoing coronavirus disease (COVID-19) pandemic is caused by the SARS-CoV-2, the RBD of the viral S-glycoprotein (S-protein) used to bind to the angiotensin-converting enzyme with low affinity -2 receptor (ACE2) to bind nanomolar range. Accordingly, S-protein is the primary target for the antibody response against the virus.
Typical antibodies have an antigen binding which is determined by the variable domains of both their heavy chain (VH) and their light chain (VL / VK); However, camelids produce unconventional antibodies only to the heavy chain, which bind to antigens based solely on the variable domain of their heavy chain – the VHH domain (also known as nanobodies).
These VHHs are increasingly used as functional antibody domains because of their small size and high stability, and VHH libraries have been screened for binders with phage and yeast displays with significant success.
In order to be able to use all advantages of the cell-free display, the developed by Dr. Xun Chen (from the Broad Institute of MIT and Harvard) led a research group on cell-free VHH identification using Clustering Analysis (or CeVICA), a novel, integrated platform for in vitro VHH-Domänen-Antikörper-Engineering.
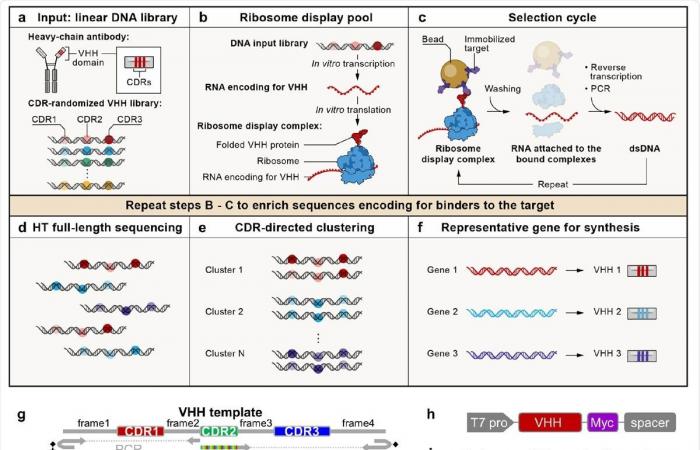
A cell-free antibody engineering platform for the rapid isolation of antibodies from large synthetic libraries. (a) The workflow uses a linear DNA library as input. (b) The ribosome display combines the genotype (RNAs transcribed from the DNA input library that are free of stop codons and block the ribosome at the end of the transcript) and the phenotype (folded VHH protein, which due to the lack of stop codons in the RNA is bound to ribosomes). . (c) The selection cycle enriches DNA encoding VHHs that bind immobilized targets. (d) High throughput sequencing of full length VHHs. (e) Sequences are grouped into clusters based on the similarity of their CDRs, with each cluster being different and representing a unique binding family. (f) The system outputs a representative sequence of each cluster to be synthesized and characterized for certain downstream applications. (g) Workflow for generating the VHH library. VHH CDR randomization was introduced by PCR using a hairpin oligo (blocks the DNA end from ligation) and an oligo with a random 5 ‘sequence, followed by orientation-controlled ligation. Three consecutive cycles of PCR and ligation randomize all three CDRs. (h) The final sequence structure of the DNA library. (i) A round of ribosome display and anti-Myc selection was performed after randomizing CDR1 and CDR2. The pie chart shows the percentage of the specified sequence categories before and after the anti-Myc selection. (j) Length distribution of the DNA region encoding CDR1 of the VHH library before and after the anti-Myc selection. Arrows indicate all correct frame lengths, which indicate an increased percentage after the Anti-Myc selection.
Optimized design
In short, this platform combines an entirely new design and generation method for randomized VHH libraries, optimized ribosome display and a selected selection cycle, and a computational approach to performing a global prediction of binders from libraries after selection.
In order to adequately conduct affinity maturation, which is a critical stage in antibody development in animals, researchers designed and implemented a CeVICA-based affinity maturation strategy to increase the affinity of RBD-binding VHHs.
They then calculated their amino acid profiles in relation to position and determined the change in each amino acid fraction at each position for each VHH – which in turn creates a percentage point change table.
The researchers also defined putative beneficial mutations and compiled a list of them for each VHH and included their various combinations in each VHH parent sequence to generate multiple mutant variants for final evaluation.
Finally, they investigated the possible effects of these VHH sequences on immunogenicity in humans, since a major problem associated with the therapeutic use of VHH antibodies is the possibility that, as camelid proteins, they would elicit an immune response.
Efficient and careful prediction of binders
Of 14 binders tested experimentally, six showed an inhibition of infection with the pseudotyped SARS-CoV-2 S lentivirus. The maturation of antibody affinity also increased the binding affinity as well as the effectiveness of the inhibition.
In addition, CeVICA’s strong ability to predict binders efficiently and comprehensively allowed the retrospective validation of the suitability of the VHH synthetic library design shown in this study as well as uncovering the direction for future refinements.
The data obtained also imply that at least some of the VHH brand residues can be converted to human residues without losing binding fitness. These conversions can then serve as framework features for future VHH library designs and then improve tolerance in vitro constructed VHHs by humans
A viable strategy for refining antibodies
Taking all factors into account, the affinity maturation expansion of CeVICA provides a viable strategy for refining antibody function, while additional iterations of the affinity maturation process can result in additional improvements in antibody properties.
“With CeVICA we have generated a large collection of antibodies that can bind the RBD domain of the SARS-CoV-2 spike protein and neutralize the pseudotyped virus infection, thus providing an important resource,” emphasize the study’s authors bioRxiv Paper.
In any case, such a harmoniously integrated process opens the door to automation and could be an important tool for fast, scalable and reliable antibody generation. A CeVICA technology framework can overcome limitations in vivo Suitability of antibodies generated in the laboratory, but also overall efficiency, which must be proven by further studies.
* Important NOTE
bioRxiv publishes preliminary scientific reports that are not peer-reviewed and should therefore not be considered conclusive, guide clinical practice / health-related behavior, or are treated as established information.
These were the details of the news Rapid generation of divergent synthetic antibodies against SARS-CoV-2 in a cell-free... for this day. We hope that we have succeeded by giving you the full details and information. To follow all our news, you can subscribe to the alerts system or to one of our different systems to provide you with all that is new.
It is also worth noting that the original news has been published and is available at de24.news and the editorial team at AlKhaleej Today has confirmed it and it has been modified, and it may have been completely transferred or quoted from it and you can read and follow this news from its main source.