The rapid spread of the COVID-19 pandemic caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has led to tremendous efforts to contain the disease. The virus’ spike protein plays a key role in infection and disease progression. The spike protein binds to the human angiotensin converting enzyme 2 (ACE2), facilitates entry into the host cell and causes infection.
The SARS-CoV-2 virus is a single-stranded positive sense RNA virus. It has a genome size of 26 to 32 kb. The genome encodes the four main structural proteins, the spike, nucleocapsid, envelope and membrane proteins. The envelope and membrane proteins are related to the virus assembly.
The spike protein interacts with the peptidase domain of the ACE2 receptor to help the virus enter the host cell. Preventing this interaction can therefore potentially be useful in containing the infection.
Aside from vaccine development, much effort has focused on drugs and therapies to limit early-stage infection. These include antibodies from blood plasma in convalescent patients, the reuse of existing drugs, and the design of blocking peptides. Peptides are attractive for combating viral infections because they are close to natural peptide conformations.
Published in a new study in the journal Cellular and Molecular BioengineeringResearchers report peptide derivatives or peptidomimetics based on the circle residues involved in the interaction between the spike protein and ACE2.
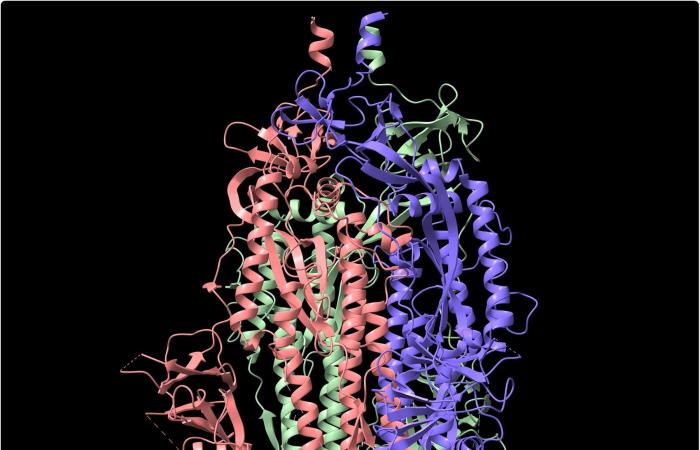
Structure of the SARS-CoV-2 spike glycoprotein, black background, 3D image credits: Volodymyr Dvornyk / Shutterstock
New peptide derivatives
Targeting peptides are advantageous because they have good structural compatibility with target proteins and can disrupt protein-protein interfaces.
The researchers had previously developed a peptide with 18 amino acids (18aa) for SARS-CoV-2 inhibition. In order to produce this peptide, they first received the crystal structure of the SARS-CoV-2-ACE2 complex and examined the residues involved in the interaction.
They found that a portion of the N-terminal region of the ACE2 peptidase domain was the major part that interacted with the virus spike protein.
Then they designed new peptide derivatives based on the inhibitory residues on the 18aa peptides. These peptide derivatives can bind to the receptor binding domain (RBD) of the spike protein better than the peptide.
Using the pep: MMs: MIMIC server, they obtained about 200 conformations of the peptidomimetics. They performed a virtual screening of these 200 connections with the HVTS (Hig-Throughput Virtual Screening) using the LibDock platform of the BIOVIA Discovery Studio.
The team then analyzed the compounds obtained using a virtual molecular docking-based screen to find the best compounds that could stably bind to the spike protein. They looked for connections with the best docking pose, and the interactions were similar to those of the binding sites of the 18aa peptide.
They obtained ratings for all compounds by comparing the uniformity of the mode of binding and the energy rating pattern of the final peptide derivative residues. From this list, they selected the four best compounds for further analysis.
Four Promising Compounds Against Spike Protein
The researchers further analyzed how each peptide derivative interacted with the spike protein residues. All four compounds showed a strong interaction and had noncovalent bonds such as hydrogen bonds, pp interactions, and cation-p interactions. These non-covalent bonds stabilize the protein-ligand complex as well as the dynamics and thermodynamics of the system.
Further analysis showed that the aliphatic chain regions attached to the aryl rings are important for hydrogen bonding and the pp interactions with the spike protein.
None of the four compounds were toxic as analyzed using the ‘ToxinPred’ server. The compounds were less lipophilic and had higher solubility. Computer simulations showed that these compounds have good binding affinity and suggest good potential for drug use.
The analysis of the molecular electrostatic potential showed the most electropositive and most electronegative areas in the four compounds. In the compounds, the electropositive areas were in the areas with amine groups and the electropositive area in the areas with protonated groups and carbonyl groups.
The amount of peptides unbound in plasma provides an understanding of pharmacokinetic properties and helps screen candidates early in drug discovery. The unbound fractions can bind to proteins and form complexes.
The four compounds have a predicted unbound fraction between 0.4 and 0.6. The total clearance value of these compounds, which is a combination of renal and hepatic clearance, indicates the adaptability of their physiological environment.
A maximum recommended tolerated dose of 0.48, which estimates the toxic dose threshold in humans, is considered low. The value obtained for these compounds is about 0.39, so they are non-toxic to humans. Thus, these compounds can be further tested experimentally in SARS-CoV-2 spike protein inhibition tests.
These were the details of the news New peptide derivatives that target the SARS-CoV-2 spike protein for this day. We hope that we have succeeded by giving you the full details and information. To follow all our news, you can subscribe to the alerts system or to one of our different systems to provide you with all that is new.
It is also worth noting that the original news has been published and is available at de24.news and the editorial team at AlKhaleej Today has confirmed it and it has been modified, and it may have been completely transferred or quoted from it and you can read and follow this news from its main source.